Making clinical research crystal clear
KrystelisDisclosureAI
AI-support for clinical trial registrations and results
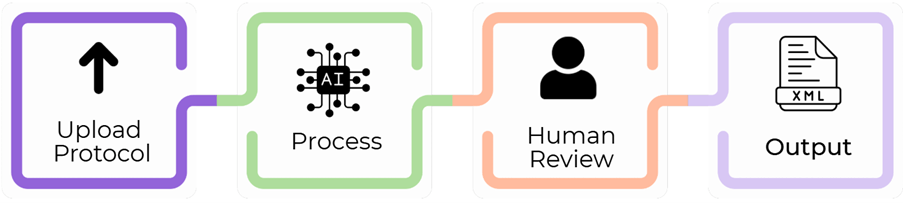
Registration and results disclosure of clinical trials on ClinicalTrials.gov is a critical regulatory requirement for sponsors, but the process is complex and can be time-consuming. The Krystelis team has prepared hundreds of registrations and results records for ClinicalTrials.gov. We have used this experience to develop an AI-powered solution to simplify, standardise, and accelerate the generation of compliant trial registration and results records for ClinicalTrials.gov.
Within minutes, the tool uses the source documents to create high-quality registration and results records consistent with the registry requirements. It also seamlessly transforms the generated record into ready-to-use XML format. This significantly reduces the effort and time required to complete the submission process.
KrystelisDisclosureAI is hosted in a secure environment to protect your data at each stage of the process and is compliant with GDPR and HIPAA regulations.

Key Benefits
Develops ClinicalTrials.gov registration form from the clinical trial protocol
XML generation for direct upload to the PRS database
Accelerates the trial registration process
Lower cost than a manual approach


